Unexpected HCV donor-derived transmissions on the rise
Published on: Saturday, March 9, 2019
Although the opiate epidemic in the U.S. is geographically variable, every OPTN region has been impacted (Fig. 2, 3). Nationally, this is one reason more transplants, especially those from donors that could place the potential recipient at increased risk of disease transmission, are being completed (Fig. 1).
Multiple manuscripts have been published that detail the successful use of organs from donors who are currently using and abusing drugs. However, challenges remain and clinicians and patients continue to be cautious, especially regarding the risks of inadvertently acquiring viral hepatitis and/or HIV. Fortunately, hepatitis B remains relatively uncommon in the U.S. and unanticipated HIV transmissions have not been documented since the new Public Health Service (PHS) Increased Risk Guidelines were put into place in 2013. However, unexpected hepatitis C (HCV) donor-derived transmissions have been on the rise. The OPTN, and specifically the OPTN Ad Hoc Disease Transmission Advisory Committee (DTAC), have reviewed these cases and made a number of observations.
Figure 1. Number of Deceased Donors Recovered by Year and PHS Increased Risk Status, 2008 – 2018.
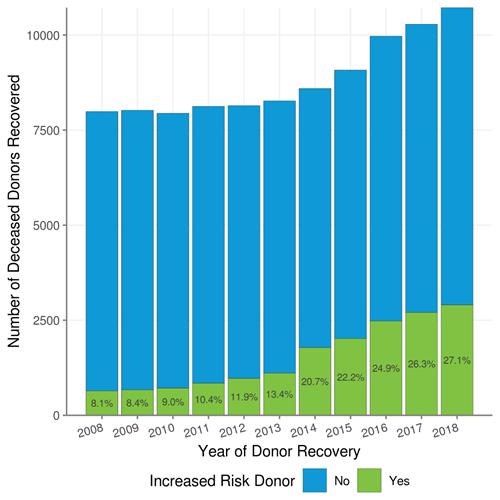
Figure 2. Proportion of Deceased Donors With a History of IV Drug Use and
Death Due to Drug Overdose During 2008 and 2018, by Region.
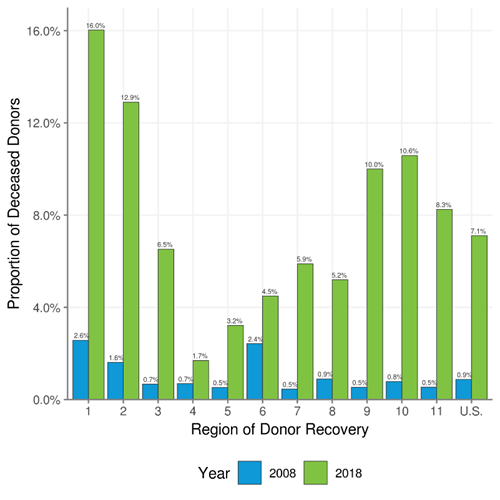
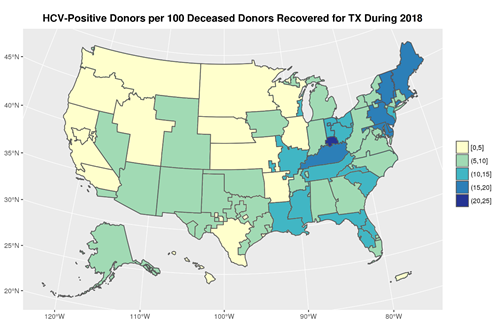
Figure 3. Number of HCV-Positive Donors per 100 Deceased Donors
Recovered for Transplant, by Donation Service Area During 2018.
HCV seropositive, yet non-viremic (Ab+/NAT-) donors
Among donors who are HCV seropositive, yet non-viremic (Ab+/NAT-), the only cases of HCV transmission occurred from donors actively using drugs. We recognize that a past history of HCV affords no protection against reinfection, especially if a donor is still actively using intravenous drugs.
Consequently, the risk of HCV transmission in these donors should probably be based solely on their behavioural risks—the same as their HCV seronegative donor counterparts. This is supported by recent publications from clinicians in thoracic and abdominal transplant programs across the country.
Donor-derived HCV
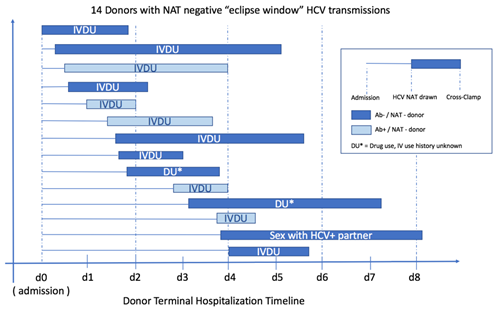
Similarly, most cases of donor-derived HCV in the past 5 years have been associated with donors among whom active drug use was recognized; most were IVDU (Fig 4). Consequently, there exists significant stratification of risk, even within the binary PHS Increased Risk framework. If a donor is an active intravenous drug user, their risk of acute HCV infection is real, and meaningfully higher than almost any other behavioral risk factor.
Figure 4. Unanticipated donor-derived transmissions, 2013-2017
Donor serology and NAT testing
Finally, an important trend in donor evaluation is emerging, whereby to ensure timely donor testing, and organ placement, many OPOs are now running donor serology and NAT testing sooner, even on the day the patient arrives in hospital. While this efficiency can be beneficial, we should view negative hepatitis or HIV tests cautiously. The NAT window for HCV is still 3-5 days, and for HIV 5-6 days. Re-exposure within that window is possible and will not be detected by any test.
Ultimately, this data should be balanced against the recognition that understanding of the safety and efficacy of HCV treatments in transplant is improving day by day. The growth of intentionally mismatched NAT+ HCV transplants speaks to our rapidly changing horizon. DTAC believes OPTN data data can help centers better adjust to the ongoing opiate epidemic, improve their consent process with patients, and ultimately maximize safe organ utilization.
For more information, refer to the DTAC guidance document available here.



