Changes to informed consent requirements for potential living donors
View commentsProposal Overview
Status: Implemented
Sponsoring Committee: Living Donor
Strategic Goal: Improve waitlisted patient, living donor, and transplant recipient outcomes
Effective date: 6/1/2017
Patient and professional resources 6/2017
Policy notice 1/2017 (PDF)
Board briefing 12/2016 (PDF - 268 K)
UNOS and OPTN member impact summary 12/2016 (PDF - 861 K)
Executive Summary
In February 2013, the OPTN/UNOS implemented the current requirements for the informed consent of living kidney donors. Informed consent requirements for living liver, pancreas, intestine and lung donors followed in 2014. Since initial implementation, several developments support the need to update and clarify the current informed consent policy requirements including:
- Publication of new evidence on living kidney donor health outcomes
- Consensus-based recommendations from professional societies that the new information regarding the health outcomes for living kidney donors should be disclosed as part of the informed consent process
- Release of living donor program-specific reports (PSRs) by the Scientific Registry of Transplant Recipients (SRTR)
- Reports from living donor program site surveys identifying areas of existing policy language that have been frequently misunderstood by living donor recovery programs
The OPTN/UNOS Living Donor Committee (Committee) reviewed the informed consent requirements for living donors and proposes clarifying existing requirements and adding other new requirements. The proposal also includes related changes to the informed consent requirements for kidney paired donation, and modification and elimination of some related OPTN Bylaws requirements.
This is a goal 3 project under the OPTN Strategic Plan to improve waitlisted patient, living donor, and transplant recipient outcomes.
Read the full proposal. (PDF - 246 K)
Feedback requested
- The Committee is interested in feedback concerning whether there are other informed consent processes or consent elements that should be considered as requirements for living kidney, liver, lung, pancreas or intestine donors but are not addressed in current policy or in these proposed changes.
- The Committee is requesting specific feedback on whether elements of the proposal would be problematic for members to implement. Feedback is especially encouraged from living lung, intestine, and pancreas donor programs because of the low volume of these types of live donations.
For any identified problem, please provide potential solutions for the Committee to consider.
UNOS and OPTN member impact summary
Project size/Complexity
UNOS
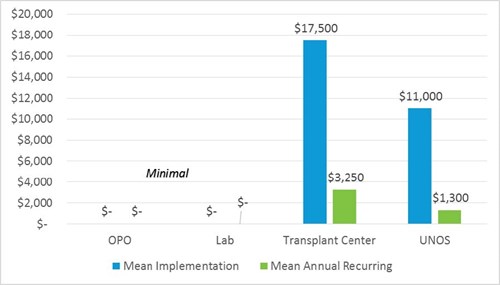
The majority of hours (80) is required from IT to program changes. Communications and Regional Administration each estimate 20 hours to communicate changes to members.
Implementation and ongoing effort is minimal.
Project size = Very Small
Member
Transplant center
Implementation timeframe for Transplant Centers is estimated at six months for most centers. Time is needed primarily to conduct legal review, implement process and workflow changes, and create print materials and to educate existing staff and patients on new requirements.
Implementation cost to transplant centers results from additional staff hours. The legal review team conducts analysis and approves new requirements, project management staff implements changes, and administrative and clinical staff educates existing staff and patients on new requirements. Printing costs exist, as well, but may be minimal. Cost and financial burden varies based on size of transplant program.
Transplant center methodology notes
- One-time implementation cost impact estimate (for most transplant centers) of $10,000-$25,000 is based on 200-500 hours of staff implementation time at a blended rate of
$50/per hour. - Recurring annual additional budget cost post-implementation ranges from $1,500-$5,000 for annual legal team review of policy requirements and printing. Cost depends on
size/volume of transplant program. - There is no cost savings.
OPOs and Labs
OPOs and labs are not affected.



